|
Again, the mass of a mole substance is called the molar mass, and to find the molar mass of a substance you just need a periodic table and the chemical formula. A solution that is 1.00 molar (written 1.00 M) contains 1.00 mole of solute for every liter of solution. That’s the total molar mass of the compound CO2. The most common way to express solution concentration is molarity (M), which is defined as the amount of solute in moles divided by the volume of solution in liters: M moles of solute/liters of solution. Then finally you add all products, so 12 plus 32 is 44. You would multiply it by its subscript of 2, which is 32. Then you’d do the same thing with oxygen. Molar mass for CO 2 molecules 1 x (molar mass of C atoms) 1 x (12. 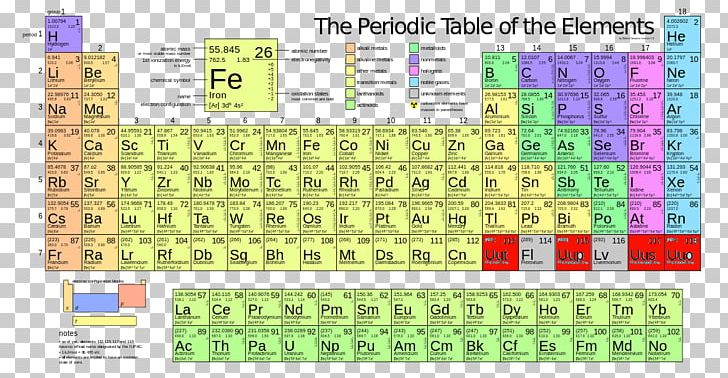
Visualize trends, 3D orbitals, isotopes, and mix compounds. Molar mass for O 2 molecules 2 x (molar mass of O atoms) 2 x (16.00 g/mol) 32.00 g/mol. 1 Chemistry Learning Laboratory The Mole, Molar Masses, Molarity, and Molar Conversions MASTERY MAP: Track your progress Learning Outcome Typical Test Questions I can do it on my own I can do it after getting help I am still confused after getting help Calculate the molar mass of an element or compound. Then the way you would find that is first you would find the atomic mass of carbon, which is 12, and then you’d multiply it by the subscript, which is just an understood 1 right there, so the product there would be 12. Interactive periodic table showing names, electrons, and oxidation states. If you were trying to find the molar mass of the compound carbon dioxide, which looks like this-CO2-and the molar mass is 44. Then, to find the molar mass of the compound, you’re going to add all those products together. By multiplying the atomic mass by the subscript you’re getting the product, you’re getting the molar mass product for that atom. That way, you’re finding the total molar mass for all of that atom because you’re finding the atomic mass, then you’re multiplying it by however many times it appears in the compound. After you’ve found the atomic mass, multiply the atomic mass by the subscript. You’re going to start with one atom at a time. I’m going to show you a 3-step process for finding the molar mass of a compound.įirst, find the atomic mass of an atom. You kind of follow the same protocol for finding the molar mass of an element, but then you combine all those molar masses together to find the molar mass of the compound. The representative particle of CO2 CO 2 is the molecule, while for Na2S Na 2 S it is the formula unit. In both cases, that is the mass of 6.02 ×1023 6.02 × 10 23 representative particles. Now the molar mass of a compound can be calculated by summing the molar masses of each atom and the chemical formula. The molar masses are 44.01g/mol 44.01 g/mol and 78.04g/mol 78.04 g/mol respectively. That’s how you find the molar mass of an element. Now we’ll write it with a different unit because it’s also our molar mass, and that will be grams over moles: \(\frac\). If you’re looking at the element of copper you would look in the periodic table and find that it has an average atomic mass of 63.55 AMU, so that’s the molar mass. If you’re trying to find the molar mass of an element, then you’ll take the average atomic mass listed in the periodic table. To calculate the molar mass of a substance you need a periodic table and the chemical formula. For instance, the relative atomic mass of zirconium (Zr) is 91.22. This is usually located under the symbol and name of the element. To find an element’s molar mass, start by checking the periodic table for the relative atomic mass of the element.  
For a molecule or compound, simply add up all the molar masses of the elements, taking subscripts into account. Molar mass is the mass in grams of 1 mole of any given substance. A mole of 12C weighs 12 g (its molar mass is 12 g/mol).The mass of a mole of substance is called the molar mass. The molar mass of any element is on the periodic table. 
Per the amu definition, a single 12C atom weighs 12 amu (its atomic mass is 12 amu). The molar mass of any substance is numerically equivalent to its atomic or formula weight in amu. (credit: modification of work by Mark Ott) These weights are known as the atomic mass, which is a number you can usually find below each element in the periodic table. From left to right (bottom row): 32.1 g sulfur, 28.1 g silicon, 207 g lead, and 118.7 g tin. From left to right (top row): 65.4 g zinc, 12.0 g carbon, 24.3 g magnesium, and 63.5 g copper. The molar mass is used to convert grams of a substance to moles and is used often in chemistry. \): Each sample contains 6.022 10 23 atoms -1.00 mol of atoms. The mass of a mole of substance is called the molar mass of that substance.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
